Discovery Engine
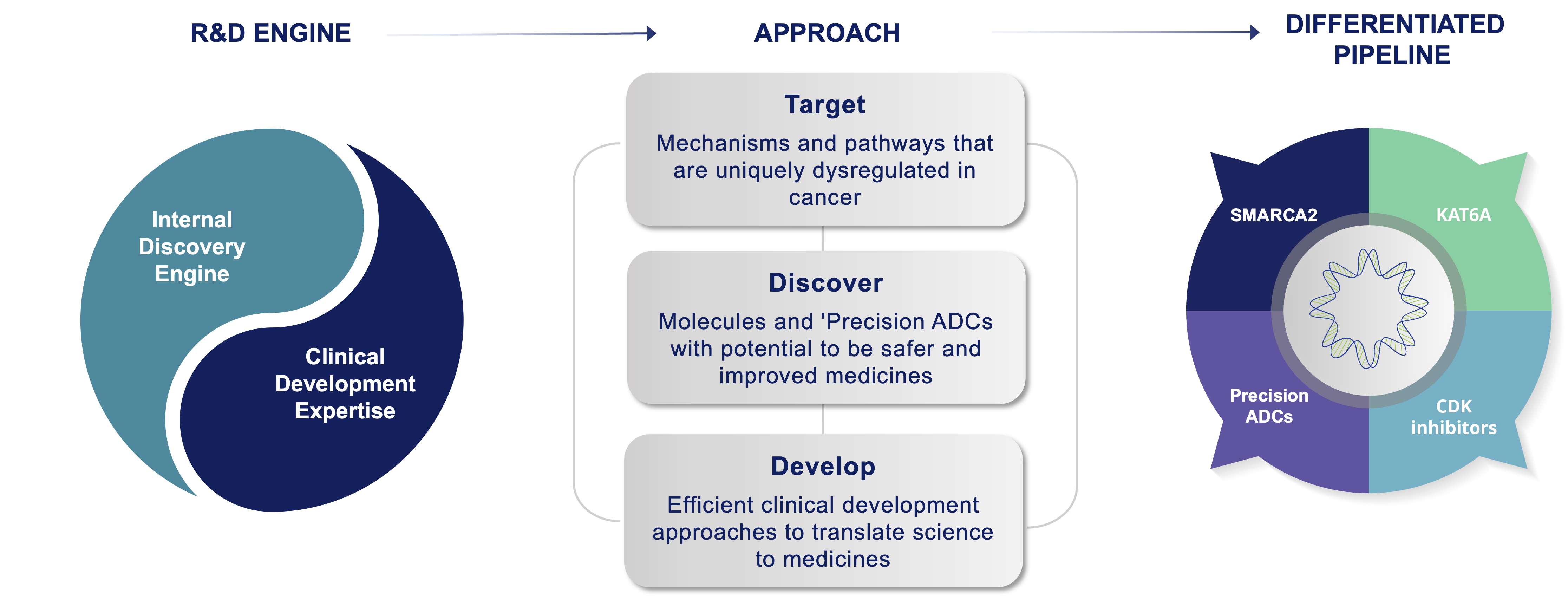
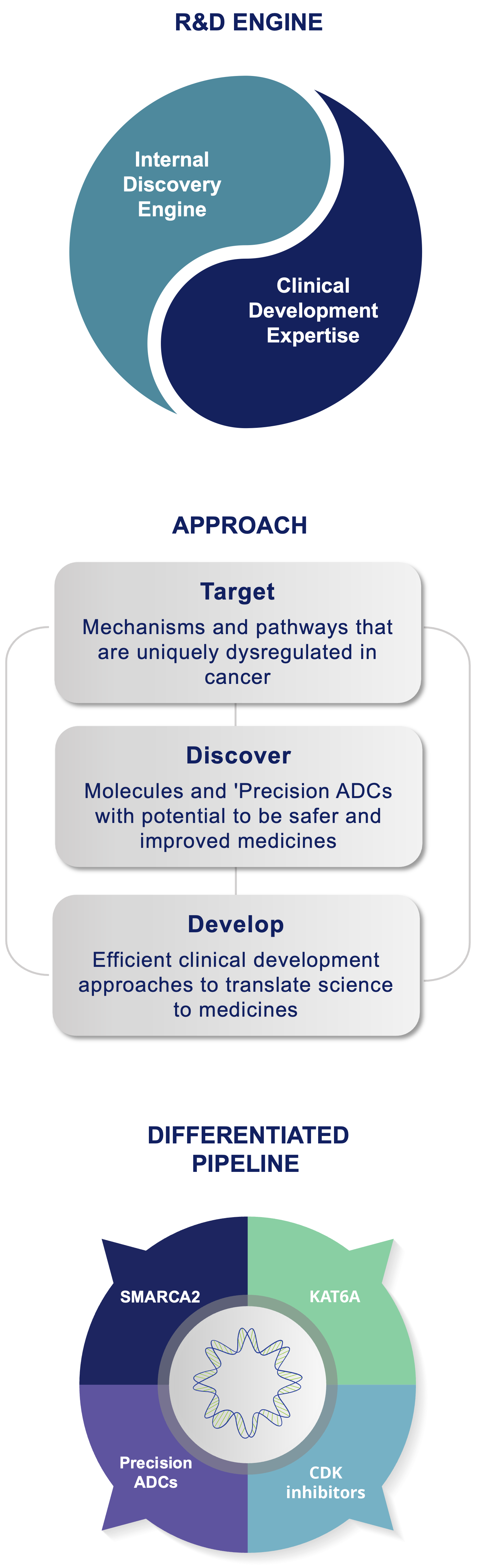
The Prelude Drug Discovery Engine has been purpose-built to accelerate the iterative design/synthesize/test process of identifying optimal medicines that have the potential to improve the lives of patients with cancer. Our goal is to pursue deeper insights into therapeutics, while constantly challenging each of our ideas, so we can achieve better outcomes for patients.
How Do We Move So Fast from Idea to IND?
Our cancer biology team provides a roadmap for an optimal product profile and our medicinal chemists design and build molecules to meet those criteria. The team rapidly iterates the design, synthesis, and testing of typically 500-1000 novel molecules using state-of-the art modeling, chemical methodologies and automated processes over the course of 12 to 18 months. The success of our Discovery Engine is evident in our multiple clinical candidates internally discovered from idea to IND.
Prelude’s Drug Discovery and Development Approach
Our broad cancer biology and medicinal chemistry expertise across a variety of small molecule modalities, enzyme inhibitors, protein-protein disruptors, and targeted protein degraders, allows us a highly flexible approach to target selection. Thus, our focus is always on addressing the most significant unmet needs for patients with cancer and discovering and developing the highest quality clinical candidates.